2020 Director’s Challenge Awards
In fiscal year 2020, the program made five 2-year awards, ranging in amount from $200,000 to $237,000 per year. The currently funded projects, project leaders, and collaborating Institute/Centers are:
- B Cell Pilot Program for High Dimensional Analyses of Antigen-Specific B Cells
- Susan Moir in collaboration with NIAID, NIAMS, and NIDDK
- Catalyzing the NIH Pain Center
- M. Catherine Bushnell in collaboration with NCCIH, NHLBI, NIA, NIDA, NIDCR, NIMH, NINDS, NINR, and CC
- Facility for Custom Synthesis of Long RNAs with Position-Selective Labeling
- Adrian R. Ferré-D’Amaré in collaboration with NHLBI, NCI, and NIDDK
- Integrated Proteogenomic Assessments in Myeloid Malignancy Clinical Trials at the NIH
- Christopher Hourigan in collaboration with NHLBI, NCI, NHGRI, and CC
- Trans-NIH Metabolomics and Lipidomics Consortium
- Michael Fessler in collaboration with NIEHS, NCI, NEI, NHLBI, NIA, NIAID, NICHD, NIDDK, and NINDS
B Cell Pilot Program for High Dimensional Analyses of Antigen-Specific B Cells
Principal Investigator (PI)
Susan Moir, Ph.D. (NIAID) In Collaboration with NIAID, NIAMS, and NIDDK
Project Summary
The B Cell Pilot Program (BCPP) brings together a team of NIH investigators from NIAID, NIDDK and NIAMS to develop integrated platforms for high dimensional analyses of antigen-specific B-cell responses. The investigators will combine their respective expertise to develop and implement these platforms and generate toolkits and open source knowhow for the larger NIH community. The purpose of the initiative is twofold: to meet a need in the intramural program to study human B cells in health and across diseases and to develop common resources and capabilities that will provide the NIH community with tools to advance specific projects while generating databases that can be shared with others. While B cells and the antibodies they secrete are known to play an essential role in protection against pathogens, they can also be dysregulated by various conditions and become pathogenic, such as in autoimmunity and allergy, or ineffective at combating pathogens. Until recently, few tools were available to study mechanisms that lead to protective versus dysregulated B-cell responses in humans, although it has become clear with the ability to directly interrogate B-cell receptor (BCR) specificities that the human B-cell compartment is highly heterogeneous and increasingly difficult to evaluate with conventional tools. By taking advantage of new powerful technologies for single-cell analyses and multiparameter phenotyping, the BCPP will develop platforms for high-dimensional analyses of antigen-specific B cells that can be applied to a wide range of research questions in the NIH community. The first platform uses full emission spectra-based flow cytometry, an approach designed to maximize the number of parameters evaluated simultaneously and to facilitate high-dimensional data analysis, coupled with bulk and single-cell (sc) sorting capabilities to interrogate transcriptomes and clone antibodies with target BCR specificity. The second platform uses an unbiased single-cell profiling using CITE-seq, a proteomic/transcriptomic approach that combines transcriptional profiling by scRNA-seq with immunophenotyping by amplification of DNA-barcode-labeled panels of antibodies directed against cell surface proteins. CITE-seq has recently been extended to interrogate antigen-specific B-cell responses, an approach called LIBRA-seq, with barcoded protein probes that bind corresponding BCRs. The BCPP will build on recent experience with these advanced technologies to develop and implement the two platforms and design additional barcoded protein probes that will be made available to the NIH community. The goal of the BCPP is to develop and implement a transferrable experimental and computational toolkit for high-dimensional analyses of antigen-specific B cells that can be applied to any human condition or in comparative analyses of different conditions, such as vaccination, infection or autoimmunity. Collectively, these efforts will provide a roadmap to the NIH community for investigating antigen-specific B cells in different diseases of interest, including to emerging pathogens such as the coronavirus responsible for COVID-19. It is anticipated that this pilot program will also be scalable for implementation within a core facility.
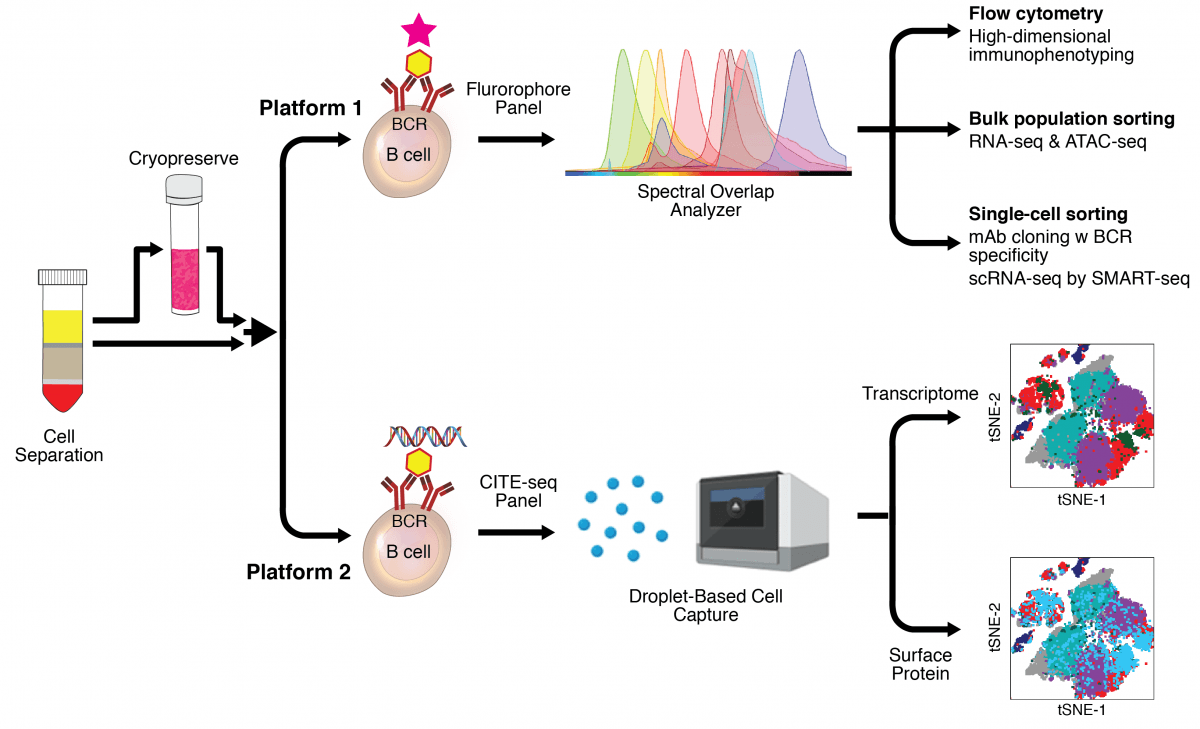
Scheme of the two platforms for interrogating antigen-specific B cells using fluorescent (Platform one) or DNA-barcoded (Platform two) probes and corresponding panels of antibodies used to perform CITE-seq and full emission spectra-based flow cytometry. Examples of specific types of applications and analyses are shown for both platforms.
Catalyzing the NIH Pain Research Center
Principal Investigator (PI)
M. Catherine Bushnell, Ph.D. (NCCIH) In Collaboration with NCCIH, NHLBI, NIA, NIDA, NIDCR, NIMH, NINDS, NINR, and CC
Project Summary
Chronic pain affects millions of people, lacks effective treatments, and is a primary driver of opioid misuse disorders. Although pain itself can be considered a disease, it is also associated with a wide range of medical conditions, including those in patients enrolled in various studies at the NIH Clinical Center. Notably, the NIH intramural program previously lacked a centralized resource to study pain within these diverse patient groups resulting in a squandered opportunity to better understand the contributing causes of pain across various diseases and identify new therapeutic targets. To address this shortcoming, a grassroots effort in the last 2 years led to the creation of a trans-NIH Pain Research Center. The Director’s Challenge innovation award will allow us to initiate staffing, training, and outfitting the NIH Pain Research Center. The mission of the Center is to create a pain phenotyping platform within the NIH Clinical Center to better understand mechanisms of diverse pain states, in order to recommend personalized therapies to better manage or prevent the development of chronic pain and opioid abuse. Three goals are to (1) identify specific pain mechanisms, (2) determine the efficacy of nonopioid treatments, and (3) predict individual patient response to therapies and outcomes. The Pain Research Center will perform deep sociodemographic, neurologic, immunologic, genetic, environmental, and biologic descriptions of individuals with pain conditions and healthy volunteers. Participants will be identified and recruited via partnerships with relevant ICs and enrolled in the multi-institute pain phenotyping protocol. Several research projects are underway, including projects tackling the central hypothesis that an abnormal balance of afferent nociceptive input and efferent modulation can produce chronic pain. With the additional funds from the Director’s Challenge, the Pain Research Center will become a nexus for intramural researchers to collaborate in deciphering mechanisms underlying pain phenotypes and transform the study of neurobiological bases of chronic pain and related conditions and pain treatment at NIH.
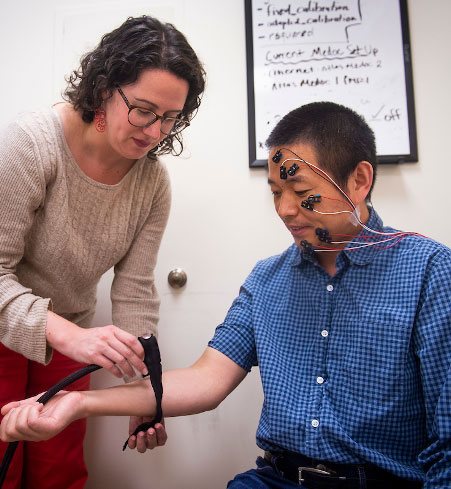
Dr. Lauren Atlas (NCCIH, NIMH, NIDA) applies a thermal heat probe to a volunteer wearing facial electromyography electrodes to measure nonverbal responses to painful stimulation.
Facility for Custom Synthesis of Long RNAs with Position-Selective Labeling
Principal Investigator (PI)
Adrian R. Ferré-D’Amaré, Ph.D. (NHLBI) In Collaboration with NHLBI, NCI, and NIDDK
Project Summary
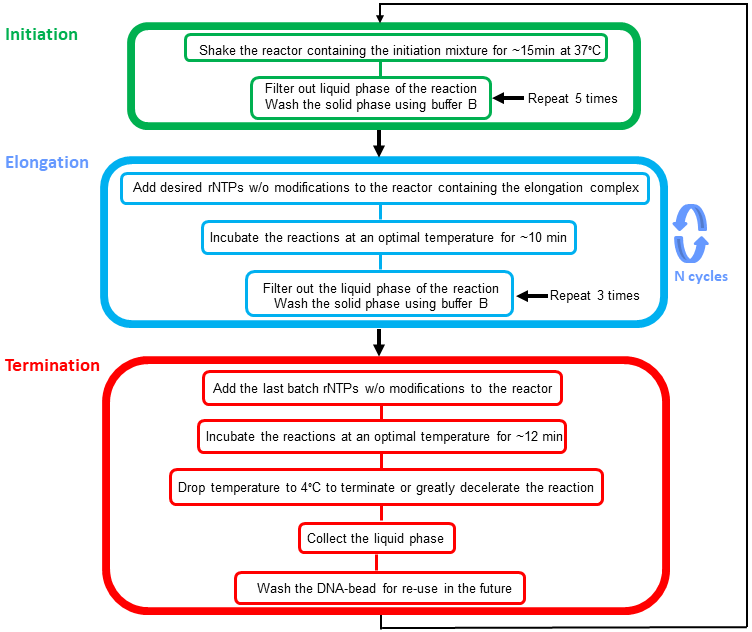
RNA plays critical roles in all areas of biology, ranging from chromatin structure to the innate immune response. The importance of RNA is reflected in the number of laboratories in the NIH Intramural Program for which it is a major focus (at least 66 labs across 13 Institutes). A key enabling technology for all experimental RNA science is the custom synthesis of RNA. Short RNAs are routinely synthesized chemically, and available commercially. What remains a major technical challenge, and is often an insurmountable obstacle, is the synthesis of longer RNAs with site-specific labels, be it fluorescent tags, isotopic, spin or nanoparticle labels, or chemical crosslinkers. This is because long RNAs are only accessible by enzymatic synthesis with RNA polymerases. While some polymerases are highly processive, and readily make RNAs that are thousands of nucleotides long, it is impossible to incorporate non-canonical nucleotides in a site-specific manner during the course of conventional in vitro transcription. Recently, the Wang laboratory (one of the four groups in this proposal) made a technological breakthrough that, in principle, overcomes this problem. The new technology achieves position-selective labeling of RNA (PLOR) by immobilizing the DNA template for transcription on a solid-phase support, and sequentially introducing nucleotide triphosphates for each step of RNA synthesis. The RNA polymerase remains bound to the template during wash steps between successive elongation cycles (Nature 52:368, 2015). This Director’s Challenge Innovation Award proposal aims to overcome several remaining practical limitations of PLOR with the ultimate goal of launching a self-sustaining PLOR core that can serve the entire NIH Intramural Program.
Integrated Proteogenomic Assessments in Myeloid Malignancy Clinical Trials at the NIH
Principal Investigator (PI)
Christopher Hourigan, M.D., D.Phil. (NHLBI) In Collaboration with NHLBI, NCI, NHGRI, and CC
Project Summary
The Cancer Genome Atlas initiative and other large-scale genetic sequencing efforts over the past decade have made clear that the blood cancers Myelodysplastic Syndrome (MDS) and Acute Myeloid Leukemia (AML) as currently clinically defined are, in fact, a heterogeneous collection of myeloid malignancies with diverse genetic etiology and the potential for intra-patient clonal evolution over time. This insight has opened up immediate clinical implications regarding diagnosis, risk stratification and treatment but have also raised more fundamental scientific questions regarding etiology and mechanisms of disease pathogenesis. There is also increasing interest in factors associated with progression from pre-malignant states (including germline predispositions and clonal hematopoiesis) to MDS/AML and the role of the immune system in causation and control of these disorders. Since 2018 a group of NIH intramural basic science and clinical investigators from across multiple institutes have been meeting to discuss ways to translate scientific knowledge generated from NIH laboratories, leveraging the unique resource of the NIH Clinical Center, to develop novel clinical trials for myeloid malignancies. This proposal builds on this momentum by establishing a new trans-NIH Myeloid Malignancies Program Clinical and Translational Correlates Facility (CTCF). The CTCF will bring together new technologies from NIH research laboratories to provide the highest quality, harmonized, cutting-edge and reproducible correlative translational science with the goal of facilitating the design, success and interpretation across the myeloid malignancy clinical trials to be conducted at the NIH Clinical Center in the coming years. While some technology would remain at the correlative research stage, it is anticipated that new clinical tests may be developed.
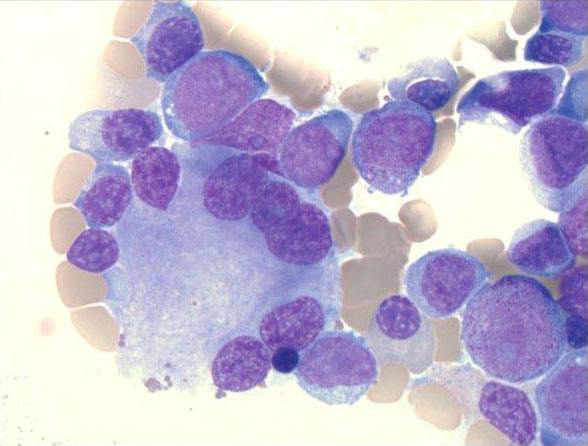
Dysplasia and myeloblasts in the bone marrow of a NIH patient with MDS/AML (Image Credit: K.Calvo).
Trans-NIH Metabolomics and Lipidomics Consortium
Principal Investigator (PI)
Michael Fessler, M.D. (NIEHS) In Collaboration with NIEHS, NCI, NEI, NHLBI, NIA, NIAID, NICHD, NIDDK, and NINDS
Project Summary
Metabolomics and the subfield of lipidomics have recently enjoyed major technical advances due in large part to developments in mass spectrometry (MS), nuclear magnetic resonance (NMR) spectroscopy, and bioinformatics. Metabolomics is currently transforming paradigms of human disease, providing new, foundational insights into cell biology as well as novel diagnostic and therapeutic leads. Unfortunately, NIH intramural metabolomics and lipidomics capability lags severely behind that of the extramural world, where the NIH Common Fund has invested heavily in a network of regional Cores and resources since 2012. Some ICs have small, but capable and well-instrumented intramural metabolomics groups. However, there is no coordination among sites, and thus no economy of scale, no standardization of methodology, and no sharing of expertise. Last year, NIEHS contacted other ICs to assess interest in creating a trans-NIH Metabolomics and Lipidomics Core Facility. After a meeting in May 2019, a survey was disseminated to intramural Investigators. Among 88 respondents across 9 ICs, there was an overwhelming degree of interest. In a subsequent meeting, governance and trans-IC service models were discussed, and mass spectrometers (and associated personnel) across the IRP were inventoried. There is a great deal of continued enthusiasm (and commitment to a sustained, long-term effort) among participating ICs, some of which, such as NIEHS, have recently made substantial investments in their local MS groups. What has been lacking, however, is the OIR-level support needed to catalyze steps forward by formalizing this consortium and launching it with committed, trans-IC resources. In this application, we propose to cement the organizational foundations for a Trans-NIH Metabolomics and Lipidomics Consortium and to initiate a developmental phase towards creation of a Trans-NIH Metabolomics and Lipidomics Core Facility. To this end, we propose the following four Specific Aims: (1) Central staffing with a coordinator; (2) Creation of a central repository of chemical and MS reference libraries; (3) Design/maintenance of a consortium website that will catalogue IRP instrumentation/personnel and post SOPs, links to extramural resources, and upcoming training events; and (4) Establishment of a bimonthly metabolomics seminar series and annual scientific meeting for consortium members and other stakeholders. A trans-IC Advisory Committee will also guide the developmental phase of Core operation using a ‘hub-and-spoke’ model (i.e., a central coordinator and distributed MS groups performing distinct metabolite panels). We are confident that this proposal describes a high-yield investment for OIR that addresses a major unmet need in the IRP.
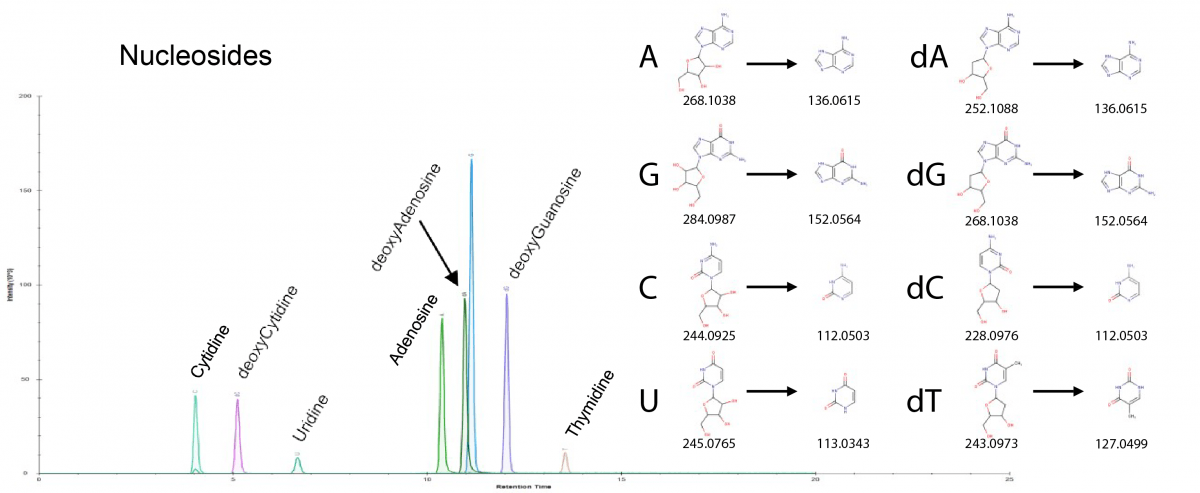
The figure depicts ion chromatograms for nucleosides and deoxynucleosides. Shown at right are m/z values for the (deoxy)nucleosides and for their nitrogenous base fragments.
This page was last updated on Thursday, July 28, 2022